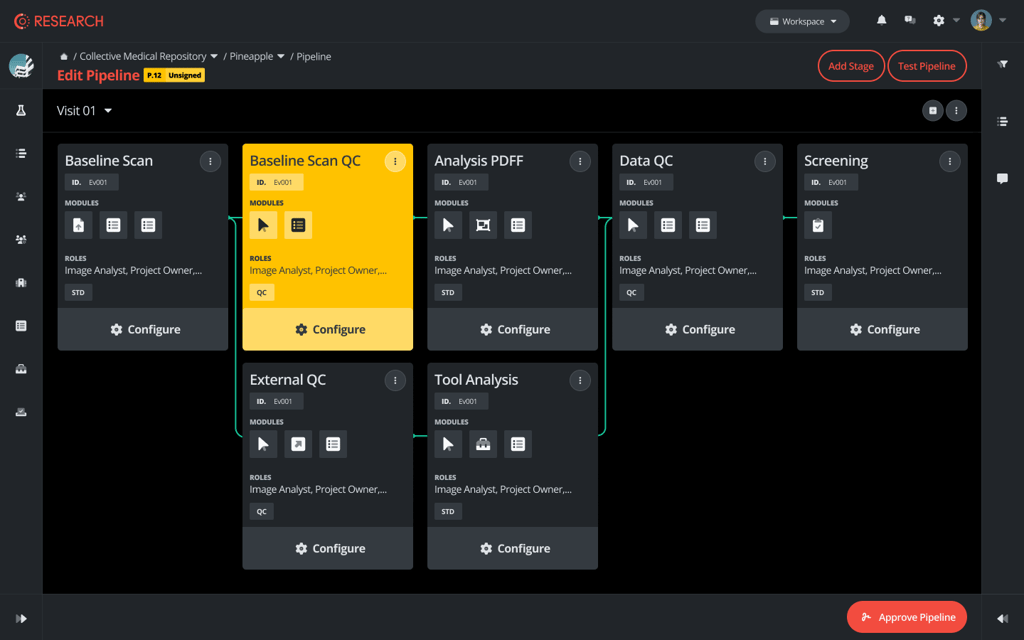
Clinical Trial Imaging Platform
- GCP
- DICOM de-identification
- HIPAA/GDPR/21CFRP.11








How it works

Collect
Send DICOM images from your Scanner or PACS, manually or via automation, directly to Connect, all within your secure hospital network.

Pseudonymize and De-identify
Imaging data is processed locally and de-identified, anonymized, and/or pseudonymized using rules you control.

Validate
Standardized QC checks flag artifacts, missing series, and protocol deviations in minutes; site can re-image before the participant leaves.
Review
Assign cases to blinded readers, run BICR with adjudication path, and capture structured assessments inside built-in eCRFs.

Track
Live trial dashboards show site progress, query rates, and reader status across all cohorts; sponsors and CROs have a single source of truth.

Export and archive
Generate study exports for stats environments, EDC, and submissions packages, with full audit trails preserved for inspection.
Features
Role Permissions
- Assign roles such as Admin, Editor, or Viewer.
- Data protection and regulatory compliance.
- Tailor your workflow to fit your unique project needs.
- Customize each event with our reusable modules.
- Customizable Events.
Results Table
- Add new entries.
- Customize the Result view for different stakeholders.
- Store and manage different versions of your results.
- Early and partial delivery.
- Permission-based.
Reports
- Fully customizable.
- Reports progress through several lifecycle stages.
- Complete transparency: Rejected reports remain visible but unmodifiable.
- Easily sharing through email or by a link.
- Export results with just a click, saving data in CSV format.
Ensure Compliance
- Traceable via Audit Log.
- E-signature Compliance (21 CFR Part 11)
- Signed and time-stamped.
- Available Metadata.
- All actions are tracked.
Screening Reports
- Automated screening steps.
- Interim result reports mid workflow.
- Instant notifications when reports are available.
Screening Reports
- Automated screening steps.
- Interim result reports mid workflow.
- Instant notifications when reports are available.
Trusted by leading healthcare institutions









Tampere University Hospital

Oslo University Hospital

Herlev University Hospital
Integrations
Everything connects. Collective Minds integrates with the tools, systems, and workflows your team already relies on, so you spend less time on setup and more time on what matters: advancing medical research.
PACS
PACS DICOM integration via CM-Connect — a secure edge gateway that pseudonymizes and encrypts imaging data on-premise before it leaves your firewall, routing it to our Data Stage for processing.
Xnat
Seamless interoperability with XNAT research PACS. Pull annotated datasets from Collective Minds Research to your XNAT archive for further processing or long-term storage.
Single sign-on
Integrate with your hospital or university identity provider (IdP) using standard OIDC or SAML protocols for secure, single-sign-on access.
API
Automate the ingestion and orchestration of multimodal data. Perfect for high-volume clinical trials or core-lab setups.

Use cases
Clinical Trials with imaging endpoints
Simplify imaging in clinical trials with a secure, cloud-based platform that reduces costs, automates workflows, and integrates with EDC systems — delivering faster timelines, better data quality, and full compliance.
Regulatory studies and clinical trials
Collective Minds helps MedTech companies meet stringent regulatory demands with an all-in-one platform that consolidates imaging data, shortens study timelines, and supports your trial from planning through post-launch.
Multireader and multisite studies
Design and manage complex multi-site studies with ease. Collective Minds lets you assign roles, customize workflows, and give every team member a personalized task list, with full access control and real-time oversight from anywhere.
See it in action

Frequently asked questions
More questions? More answers!
What is a medical imaging platform and how is it different from a PACS?
A PACS is built for clinical reporting inside a single hospital. A medical imaging platform like Collective Minds is built to organize, share and analyze imaging across institutions and projects — adding cohort building, structured annotations, collaboration tools and audit trails that traditional PACS systems don't provide.
Is Collective Minds compliant with HIPAA and GDPR?
Yes. The platform is HIPAA- and GDPR-aligned, with role-based access control, SSO, full audit trails and configurable de-identification, so research and clinical teams can collaborate without compromising patient privacy.
Can I connect Collective Minds to my existing PACS and AI models?
Yes. Collective Minds supports DICOM ingestion and routing from existing PACS, plus APIs and integrations to plug in AI models, electronic data capture systems (such as REDCap) and analytics tools — so it fits your current stack rather than replacing it.